The family of an eight-year-old Sunshine Coast girl is petitioning the federal government to have an essential eczema medication added to the Pharmaceutical Benefits Scheme (PBS).
Charlotte Oostenbroek’s eczema was apparent at birth and she was diagnosed with severe eczema at just five weeks old. Her condition quickly became uncontrollable, eventually covering 90 per cent of her body.
Severe eczema, known medically as atopic dermatitis, affects approximately three million Australians, including one-in-three children aged six years or younger.
The broken, bleeding and intensely itchy skin affected not only Charlotte’s physical health but also her family’s day-to-day functioning, sleep and mental and emotional wellbeing.
Despite trying every available treatment, nothing brought relief. Charlotte was unable to take part in physical or outdoor activities – missing school sport, swimming lessons, swimming carnivals and beach trips.
School life was particularly difficult. Charlotte missed 42 days in one year due to repeated infections and faced daily comments from other children about her skin.
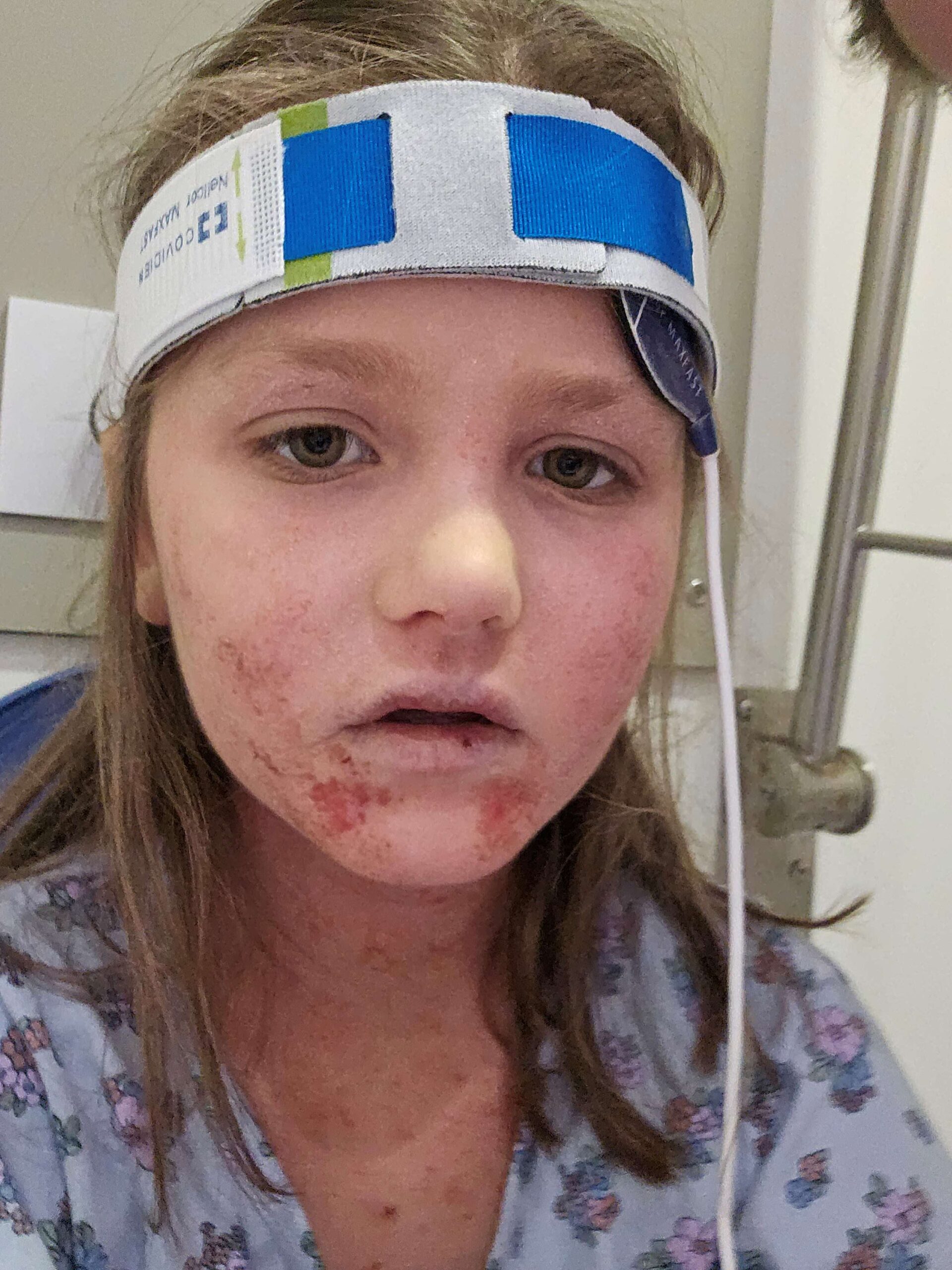
At age seven, Charlotte was granted compassionate access to a medication: dupilumab (brand name Dupixent) through Queensland Children’s Hospital. The results were transformative. Her eczema cleared completely and she has not had a single infection since starting treatment.
Charlotte is now sleeping better, gaining confidence and improving academically. She even attended her first swimming carnival.
Her family is now backing the “Save Our Skin” (SOS) campaign led by Eczema Support Australia, which is calling on the federal government to end what advocates describe as a long-standing gap in access to effective treatment for children under 12.
Dupilumab is currently available on the PBS for Australians aged 12 and older at a cost of about $25 per month. However, families with younger children must pay around $1,600 a month without the subsidy.
Help us deliver more news by registering for our FREE daily news feed. All it requires is your email at the bottom of this article.
Unlike traditional eczema treatments, which work to soothe the surface of the skin, Dupilumab targets the specific internal signals that trigger the inflammation responsible for severe eczema, providing long-term control. The injection is administered every two to four weeks and can be used by children as young as six months of age.
The campaign comes as the government considers extending PBS funding to children as young as six months old with severe eczema.
Eczema Support Australia founder Melanie Funk said thousands of children have been left behind while older patients have benefited from subsidised access for years.
“It is not right that Australian children are missing out on a treatment that has been life-changing for adolescents and adults,” she said.
“This makes no sense when we know that effective treatment in younger years can change the trajectory of a child’s life.
“It not only improves control of eczema over the long term, but also reduces their risk of related conditions, including asthma, allergies and hay fever.”
Dermatologist Dr Li-Chuen Wong is a specialist from the Australasian College of Dermatologists with extensive experience treating children with severe eczema.
Dr Wong said expanding access to dupilumab would also ease pressure on hospitals.
“We know that dupilumab works, not only to relieve the suffering of children but to save valuable hospital resources,” she said.
“Several hospitals across Australia are providing children with compassionate access to the medicine and the results have been incredible.”
Data from Queensland Children’s Hospital shows a 92 per cent drop in hospital admissions among children receiving the treatment. With each hospital admission costing an average of $8,211, advocates say broader access could deliver major savings.
Eczema Support Australia is encouraging families and medical professionals who have experience with severe eczema to share their experiences with the Pharmaceutical Benefits Advisory Committee before May 20 2026.
The Committee will meet in July before delivering a funding recommendation to the Federal Minister for Health, Disability and Ageing in August.





